Hillrom R-644-36 User manual




















Table of contents
Languages:
Other Hillrom Medical Equipment manuals

Hillrom
Hillrom A-71400 User manual

Hillrom
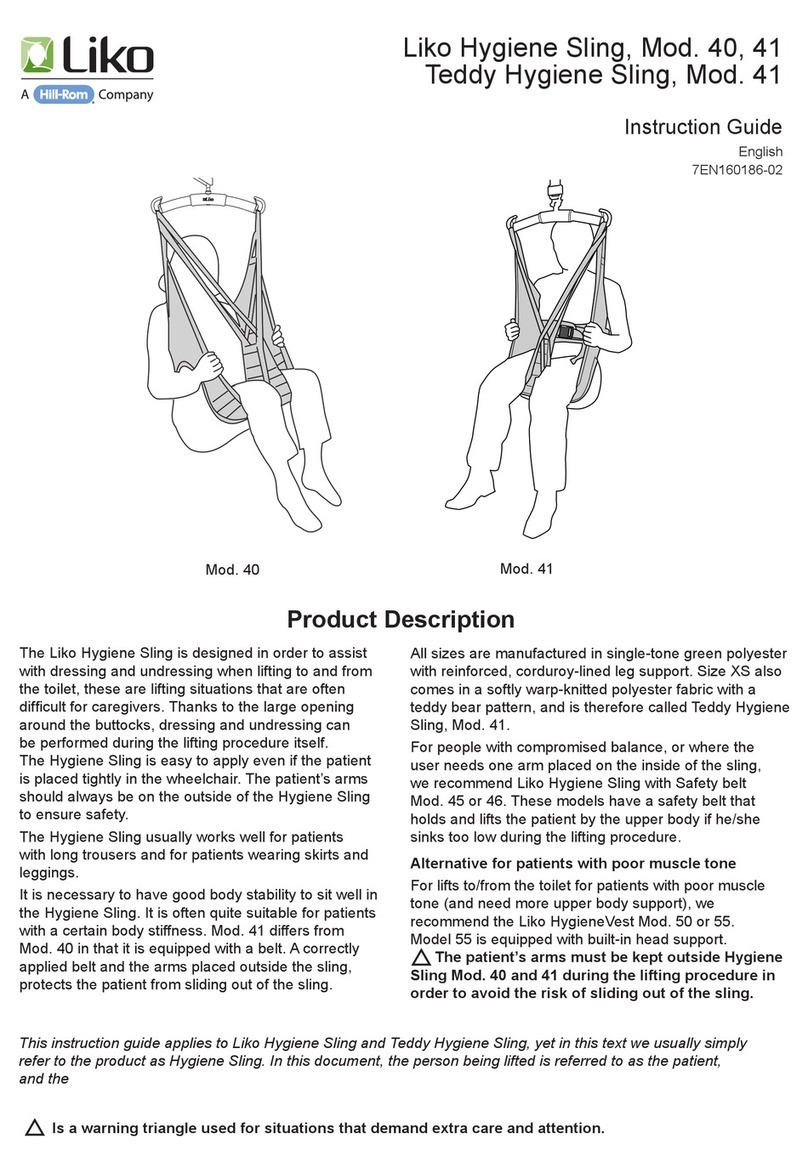
Hillrom Liko 190117 User manual
Hillrom
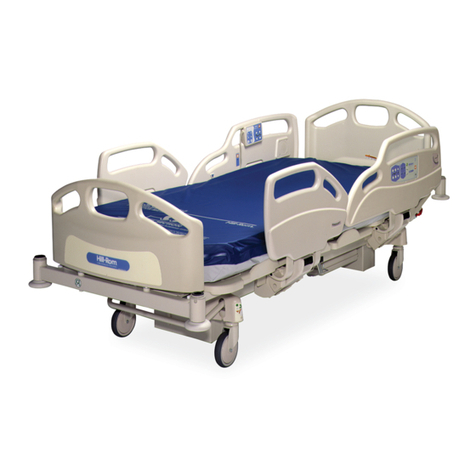
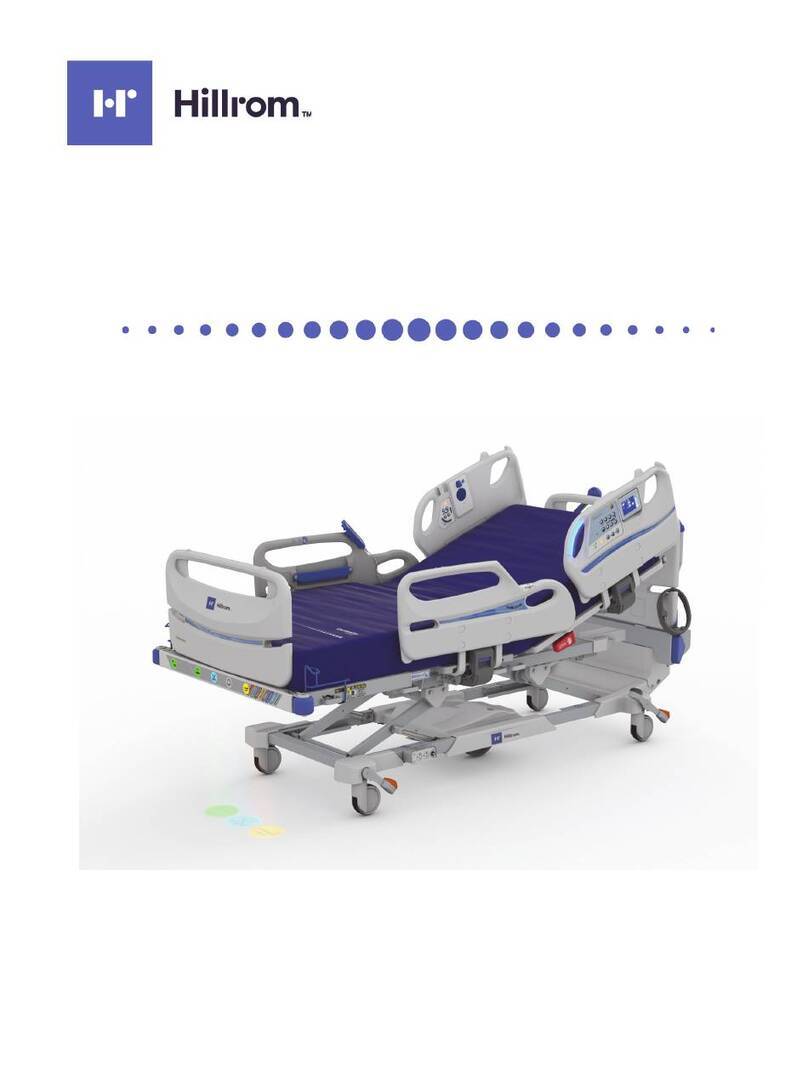
Hillrom Centrella Smart+ Bed User manual
Hillrom
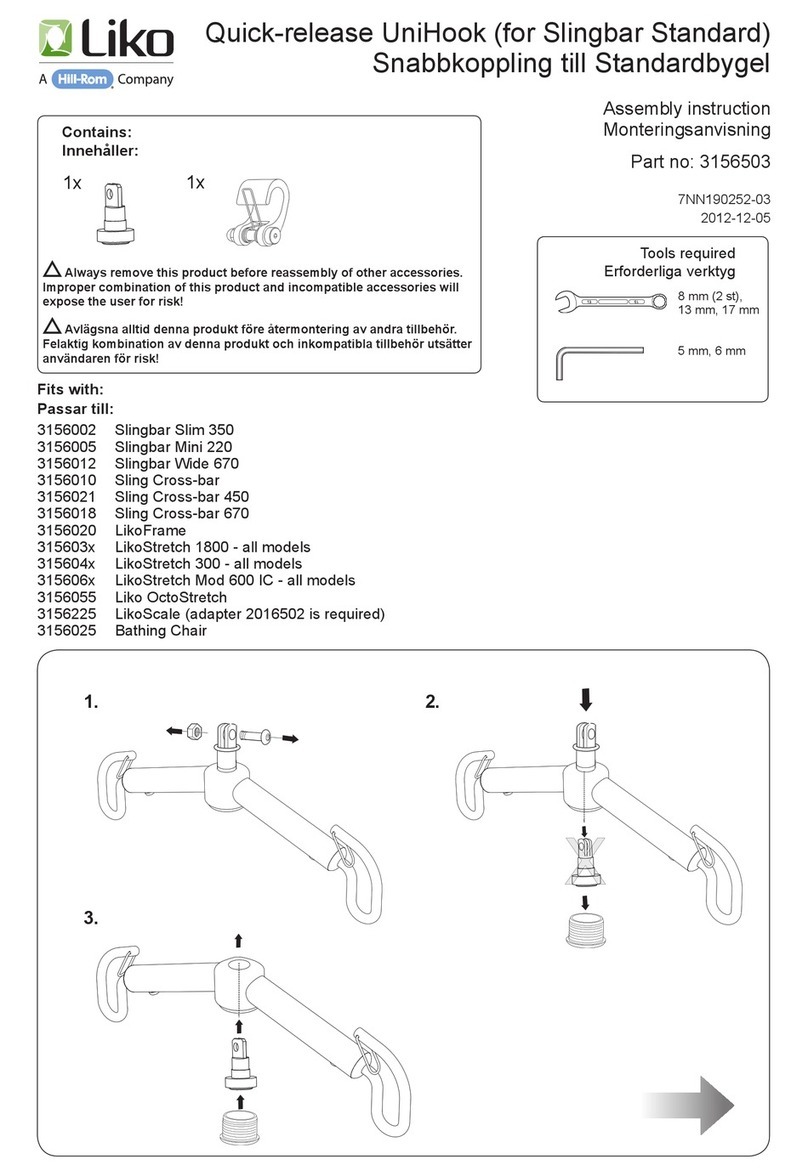
Hillrom Liko UniHook 3156503 User manual

Hillrom
Hillrom PST 500 U User manual

Hillrom
Hillrom Liko Viking M User manual
Hillrom
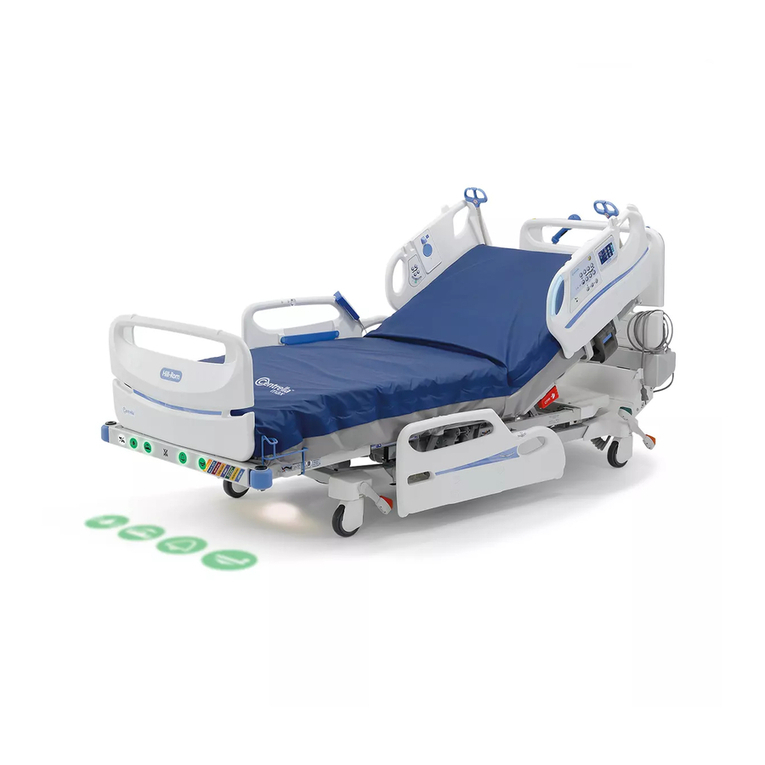
Hillrom Centrella Smart+ Bed User manual

Hillrom
Hillrom FreeStand Manual
Hillrom
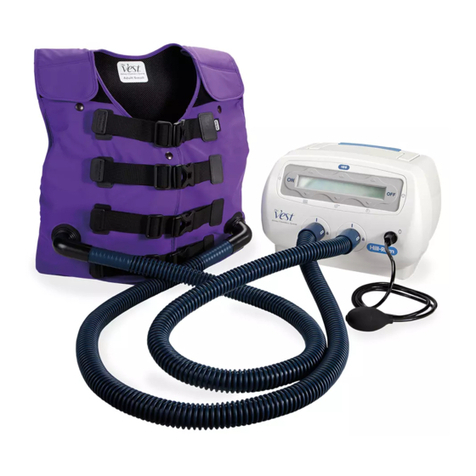
Hillrom Vest System Installation and operation manual

Hillrom
Hillrom UNIVERSAL A-90022 User manual

Hillrom
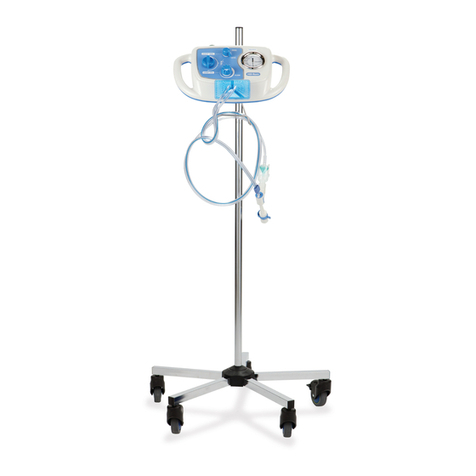
Hillrom P8000 User manual
Hillrom
Hillrom MetaNeb User manual
Hillrom
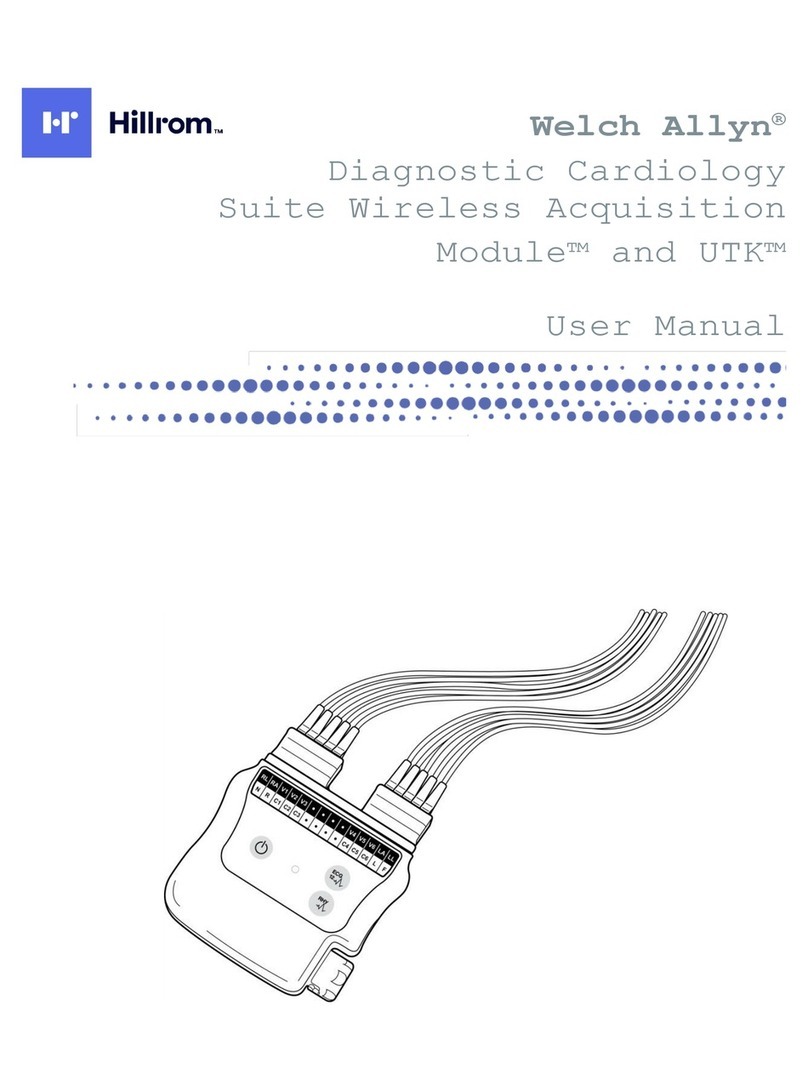
Hillrom Welch Allyn UTK User manual

Hillrom
Hillrom Basic Care P1440 User manual

Hillrom
Hillrom Allen A-70801 User manual

Hillrom
Hillrom O-ABSL User manual

Hillrom
Hillrom Liko AmputeeSling 70 Manual
Hillrom
Hillrom Welch Allyn Spot Vital Signs 4400 User manual

Hillrom
Hillrom Liko Golvo 8000 Manual
Hillrom
Hillrom Welch Allyn H12+ User manual
Popular Medical Equipment manuals by other brands

Getinge
Getinge Arjohuntleigh Nimbus 3 Professional Instructions for use

Mettler Electronics
Mettler Electronics Sonicator 730 Maintenance manual

Pressalit Care
Pressalit Care R1100 Mounting instruction

Denas MS
Denas MS DENAS-T operating manual

bort medical
bort medical ActiveColor quick guide

AccuVein
AccuVein AV400 user manual